In today’s global landscape, where medical innovation and access to strategic markets are critical challenges, Medicallians, the first international alliance dedicated to the healthcare sector, brings together local experts to support medtech and biotech companies in their international growth.
As Medicallians’ representative member in Canada, Adexia uses this article to highlight the specificities and opportunities of both the medical device and pharmaceutical markets, helping healthcare players better understand this demanding yet highly promising environment.
For several years, Canada has been attracting international players in the medical sector, particularly those specializing in medical devices. With a structured healthcare system, an aging population, and a strong commitment to independence in health technologies, the country represents a market that is both promising and demanding.
Canada at a glance :
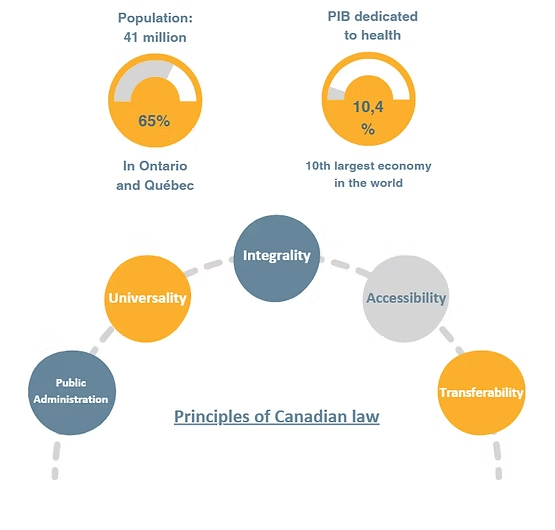
Canada provides universal access to healthcare through its publicly funded system, which is administered at the provincial level. At the federal level, Health Canada is responsible for protecting public health and setting the regulatory framework for the sector.
Provinces and territories manage and deliver most healthcare services, each with its own insurance plan, budgets, priorities, and purchasing programs. While this structure can make the market more complex for foreign companies to navigate, it also creates multiple entry points and valuable opportunities for regional partnerships.
Canada’s medical devices sector is both vast and dynamic. It brings together public and private players: hospitals, research centers, clinics, specialized distributors, and a growing network of digital health start-ups. The most in-demand products range from surgical instruments and imaging equipment to connected devices.
Supported by clusters and university-based centers, the country has fostered an environment conducive to research and R&D funding.
In this context, innovations in telemedicine, IoT-based devices, and remote monitoring tools are gaining traction, driven by the digital transformation of healthcare. The pandemic accelerated these trends while also exposing the risks of reliance on imports. This includes telemedicine, diagnostics, and AI-driven therapeutics—particularly critical given Canada’s shortage of healthcare professionals, especially in remote regions. As a result, Canadian governments are investing in local production and technological partnerships.

The Canadian medical device industry is highly diverse and not very concentrated. The vast majority of companies are small and medium-sized enterprises (SMEs). There are over 40,000 medical devices available in Canada, with about 75% of them being imported :
Entering the Canadian market requires approval from Health Canada, with devices classified according to their risk level. Unlike in Europe, CE marking alone is not sufficient: a specific process is required, sometimes involving audits, CSA/UL certifications, and bilingual labeling requirements. While these steps may slow down market entry, they provide a framework of trust for end users.
Medical devices are classified under Health Canada’s risk-based system into four categories — Class I, II, III, and IV. Licenses for Class I devices are relatively easy to obtain. Class II devices require an MDSAP certificate, while Classes III and IV involve a more complex process. However, obtaining a license in Canada is generally less time-consuming than under the EU MDR regulation.
Challenges | Solutions | Associated Opportunities |
Shortages of specialized healthcare workers, particularly in nursing, imaging, and biomedical fields | International recruitment, government incentives, increased use of telehealth and automation | Remote training, support technologies (AI, robotics), deployment of digital healthcare services |
Accessibility of equipment for people with disabilities (recurring issue, especially outside major centers) | Strengthened accessibility standards, innovation in universal design | Development of inclusive equipment, niche markets for specialized companies |
Increasing complexity of medical needs (aging population, chronic diseases, personalized care) | Interprofessional coordination, decision-support tools, integration of health data | Digital health platforms, AI for diagnostics, customized solutions |
Geographic remoteness | Telemedicine, mobile clinics, medical drones | Specialized logistics solutions, niche markets (e.g., Indigenous healthcare, northern health services) |
Need for innovation and adaptation of healthcare infrastructure | Increased public funding, PPPs, calls for innovation | Equipment modernization, smart hospital construction, sustainable healthcare buildings |
Inefficient management in some regions (persistent disparities across provinces and rural areas) | Centralization of data, best practice sharing, smart regionalization | Outsourced services, public-private partnerships, AI-driven optimization |
Shortage of mental health resources and difficulties in accessing care | Development of prevention programs, integration of mental health into primary care, support for psychological telehealth | Expansion of digital solutions (mental health apps, remote monitoring platforms), public-private collaborations, innovative therapies. |
Did you enjoy this article? Discover our other resources on international development and market trends to help you further your project in Canada!

The sector is built on a highly collaborative ecosystem:
The approval process is governed by the Food and Drugs Act and overseen by Health Canada (Health Products and Food Branch).
Other Key Points
The pharmaceutical sector in Canada remains strong, with a strong presence in Ontario and Québec. It is characterized by the dominance of multinational corporations, but also by the dynamism of local players specializing in generics and biotechnology. Despite challenges related to costs and supply, the sector offers promising opportunities in innovation, biosimilars, and local production.


The Canadian food and beverage market is mature, highly competitive, and strongly intermediated. Growth is ...

Alcohol trade regulation in Canada falls under provincial jurisdiction. Each province controls importation, distribution and, ...

SMEs and Mid-Sized Companies: Have You Considered External Growth to Accelerate Your Expansion in Canada ...

Canada is Opening the Door to Strategic Investment in Energy, Infrastructure and Critical Resources Thanks ...

Canadian engineering is entering a pivotal decade: rarely has the country mobilized so much public ...

In 2025, Ontario confirms its position as Canada’s leading economic powerhouse. With a dynamic population, ...